What is the best option for double-blind capsule preparation in clinical trials?
7 October 2016
C. Gary1, S. Djabarouti1,2, A. Venet1, G. Guyon1, P. Mora1, S. Tamisier1, F. Xuereb1,2, S. Crauste-Manciet1,3, D. Breilh1,2 1 Pharmaceutical technology department, Bordeaux University Hospital, France2 Department of Pharmacokinetics, Groupe PK/PD, INSERM U1034 - Bordeaux University Hospital, France
3 Laboratoire ARNA, ChemBioPharm, U1212 INSERM - UMR 5320 CNRS Bordeaux University, France

As part of clinical research, it is increasingly required to ensure the production of non-sterile experimental drugs. This new activity is complex and requires special agreements of ANSM. Our objective was to assess different strategies for capsule preparation for a double-blind clinical trial.
The double blind of an experimental drug consists in defining the pharmaceutical form and choosing the labelling according to the study design. We developed an analysis grid (Table 1) to compare 2 possible strategies for preparation in pharmaceutical technology department using Active Pharmaceutical Ingredient (API) (1) as raw material or as commercialized tablet (2) in an over-capsule especially designed for ensuring blindness (DBcap®). The grid was then applied for one double-blind monocentric study for which the pharmacy was asked for providing a total of 8000 capsules of verum (propranolol 20, 40 mg) and placebo.
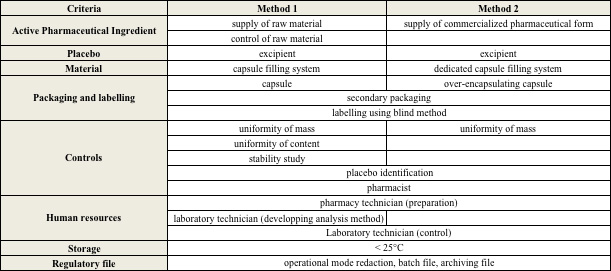
The total estimated costs were 19 500€, 33 100€ for method 1 and 2 respectively and were far lower compared to outsourcing (80 000€). For this special study, method 1 was the cheapest and this could be explained by the fact that analytical method of control was previously developed by the lab and stability data of propranolol already available. On another hand, over-capsule system needed investment in special materials (over-encapsulating capsules and special capsule filler). However, this investment could be interesting for further clinical trials involving API for which no stability study is available and needs development.
This grid will be implemented as a decision tool for further requests on preparation of capsules in clinical trials.
This work highlights the importance of pharmaceutical expertise in the choice of final pharmaceutical form in clinical research. Due to increasing implication of pharmacists, further investigation of pharmaceutical costs linked to production in clinical trials is needed.