Validation of a new peristaltic pump for batch production in isolator
7 October 2016
J. Ollivier1, C. Gary1, L. Sauvaget1, G. Bouguéon1, A. De Coucy1, V. Servant1, A. Berroneau1, D. Breilh1,2, S. Crauste Manciet1,3 1 Pharmaceutical technology department, Bordeaux University Hospital, France2 Department of Pharmacokinetics, Groupe PK/PD, INSERM U1034 - Bordeaux University Hospital, France
3 Laboratoire ARNA, ChemBioPharm, U1212 INSERM - UMR 5320 CNRS - Université de Bordeaux- Collège et département des Sciences de la Santé
To improve the quality of the process of batch preparation, we investigated the implementation of a peristaltic pump (PP) for batch production. Initials operational qualification and calibration were performed. The PP was not designed for sterilization inside isolator. The provider was able to treat surfaces of the PP with a polytetrafluoroethylene (PTFE) coating and protect all electronic connexions with plastic stoppers to allow further sterilization (peracetic acid or hydrogen peroxyde). The aim of this work was to validate the PP after PTFE coating and sterilization inside isolator.
The PP concerned is the Verderflex Vantage 3000P. To asses its use inside isolator operational qualification (OP) is required. The qualification was assessed by accuracy (A): 98%<A<102% and precision, coefficient of variation (CV < 5%). These results were compared outside and inside isolator.
A batch of 10 preparations of aciclovir 300 mg were produced using a master bag of aciclovir (5 mg/ml). Controls of batch preparation were assessed by the concentration of the master bag (analytical control) and the weight of each sub-batch bags (60.8g).
Using the original calibration, the results of OP outside and inside isolator are given in Table 1. Accuracy was not validated inside isolator.
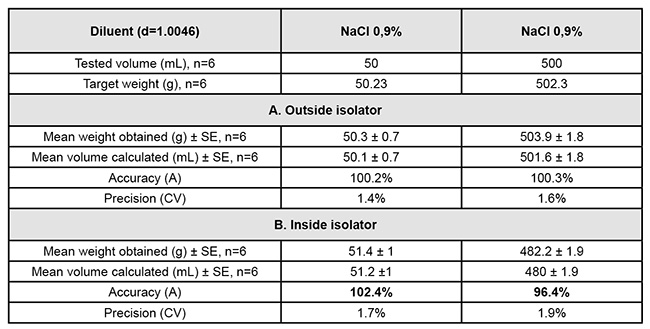
A new complete calibration inside isolator allowed to correct the accuracy (A = 100.8%) and precision was below 5% (CV=1.4%). Our results showed the impact of running the PP inside isolator showing the interest to perform a validation in real condition of use (performance qualification). A control production of aciclovir confirmed the stability of the calibration: concentration in master bag = 4.8 mg/mL (<5%) - mean (±SE) weight obtained = 59.6 ± 0.5 g (CV=0.8%), n=10.
This work highlights the impact of the isolator on calibration of the PP and confirms the need to implement performance qualification in real condition before use in routine.