Investigation into Rapid (Alternative) Microbial Detection Methods (RMM) to improve the QA of NHS manufactured aseptic products
3 October 2013
SJ. Hiom, C. Talbot, P. Spark, J. Smith, SD. Denyer SMPU, Cardiff and Vale University Health Board, NHS Wales, UK
Background
Current quality assurance is a challenge for short expiry dated, high risk aseptic products and is often based on retrospective review of environmental and product microbial monitoring. There are documented examples of quality failure with aseptic manufacture 3. RMM have been used in the Pharmaceutical and food industry for many years, early pilot work from this group with aseptic NHS products have been positive1
Purpose
This study evaluates three commercial RMM technologies for their ability to improve the traditional QA processes associated with NHS aseptic manufactured products.
Materials and Method
The three commercial RMM systems used were BacT/ALERT® (bioMerieux), AkuScreen® (Celsis) and BactiFlow ALS® (AES Chemunex). 50 µl of each of four microbes were spiked into each of four aseptic products ml (Heparin 100 u/ml; PN 7.5%; Vancomycin Inj 10 mg/2 ml and Methotrexate Inj 15 mg/0.6 ml) and left for 10 mins, producing an inoculum of approximately 10cfu/ml. All work was carried out in a Grade A environment and comparative samples were taken using split sample methodology and within 1 min of each other. Traditional methods of QA microbial recovery, TAMC as described in the BP2, were compared with results from the three RMM.
Results and Conclusion
The recovery rates and time to results are presented below as percentage concordance.
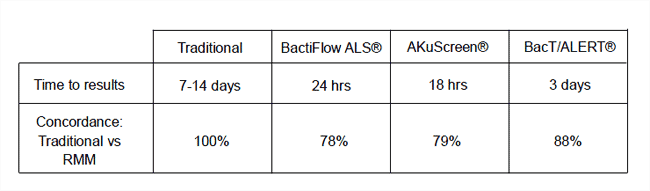
All three RMM systems were able to provide 100% concordance when used to detect contamination in PN and Heparin within their recommended time frames. All rapid methods had problems recovering G+ve organisms from vancomycin and methotrexate, although recovery of other organisms from these products were equivalent. The BacT/ALERT® system was technically the easiest to use and had highest concordance when results were read after 3 days. We believe this data describes a true reflection of the alternative methods and demonstrates that RMM can improve QA of selected NHS manufactured aseptic products.
References
- Hiom S. et al (2010). Investigation into rapid microbial detection methods (RMM) to improve the QA of NHS manufactured aseptic products. Int. J. Phar. Pract. 18 (Supplement 2) 92.
- British Pharmacopoeia. Appendix XVI B: Microbiological Examination of Non-Sterile Products. 2009. HMSO: London.
- Aseptic Dispensing for NHS Patients. Farwell report. 1995. Dept of Health
Acknowledgements
Welsh Assembly Government Pharmacy Practice Development Scheme and support from the three companies involved.