Contamination of calcium gluconate solutions by aluminum: which consequences for parenteral nutrition in neonatology?
8 October 2015
B. Raspaud, A. Venet, A. Jourand, S. Mesli, J. Grellet, D. Breilh, S. Crauste-Manciet CHU de Bordeaux, Matrice Pharmacotechnie,Pôle des produits de Santé, Bordeaux, France

Batch calcium gluconate solutions packaged in high volume glass bottles provided by AGEPS (APHP, Paris) were withdrawn because of particulate contamination. Additionally aluminum contamination was found. This contamination was partly due to the solution interaction with glass containers. Our objective was to evaluate the level of contamination by aluminum of our productions of total parenteral nutrition (TPN) bags according to the type of raw material used: AGEPS glass bottle (withdrawn batch), glass ampoule (Lavoisier®), plastic ampoule (Aguettant®) in order to assess the potential amounts which could be administered daily for each source. Raw material from different providers and bags produced from glass and plastic ampoules were analyzed by atomic absorption spectrometry.
The results are shown in the following table (table I):
Table I: Aluminum concentration and potentially administered daily amounts of aluminum
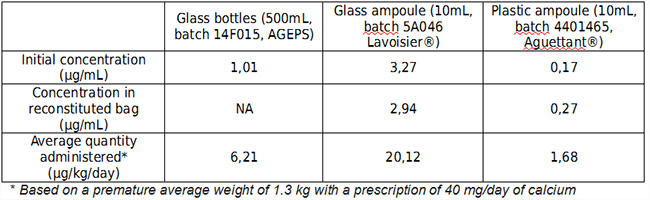
The results show a lower aluminum contamination level in TPN produced from plastic ampoules. Based on the FDA-recommended limits (<5ìg/kg/day) [1] for chronic administration, plastic ampoule (Aguettant®) is the only source acceptable with regards to aluminum contamination. However, production from ampoules introduces a significant microbiological risk (need to open 100 ampoules per day). Recently, sodium lactate solutions were subject to a similar aluminum contamination alert. A more global analysis of "aluminum risk” should be undertaken to enable an informed prescribing and integrate this potential risk.
[1] Food and drug administration. Aluminium in larger and small volume parenterals used in total parenteral nutrition Feg Reg 2000 :65 :4103-4111and amendement ; delay of effective date. Feg Reg 2002;67:70691-70692.